
Fitbit Receives Regulatory Clearance in Both the United States and Europe for ECG App to Identify Atrial Fibrillation (AFib)
Price: $ 22.99
4.8(583)
Fitbit (NYSE:FIT) has received 510(k) clearance from the U.S. Food and Drug Administration (FDA), as well as Conformité Européenne (CE) marking in the

Survey of current perspectives on consumer-available digital health devices for detecting atrial fibrillation - ScienceDirect

Fitbit Announces Fitbit Sense Smartwatch: Dives far deeper towards health metrics

Solved: When will the ECG app be available on Fitbit Sense - Fitbit Community

News - Cardiac Rhythm News

Detection of Atrial Fibrillation in a Large Population Using Wearable Devices: The Fitbit Heart Study

Fitbit Wins FDA 510(k) Clearance and CE Mark for ECG App

Fitbit ECG Apps Instruction Manual
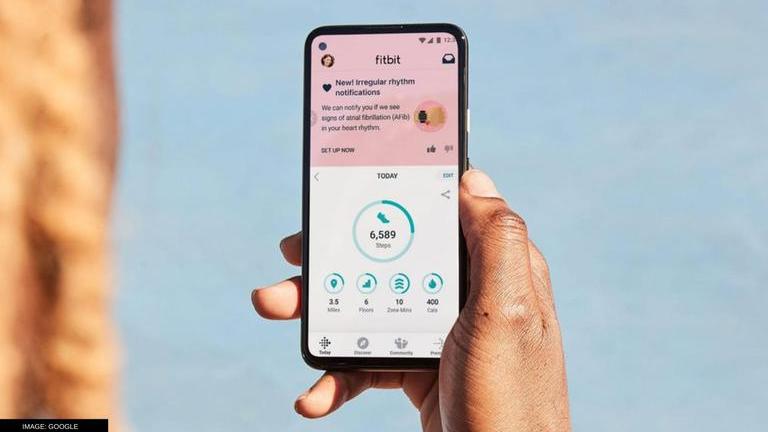
Fitbit gets FDA clearance for new PPG AFib tracking algorithm: How does it work?- Republic World

Why the new $159 Fitbit Charge 6 might be my next smartwatch

Fitbit Receives FDA Approval for a Wearable Device App That Detects Atrial Fibrillation - Dark Daily




